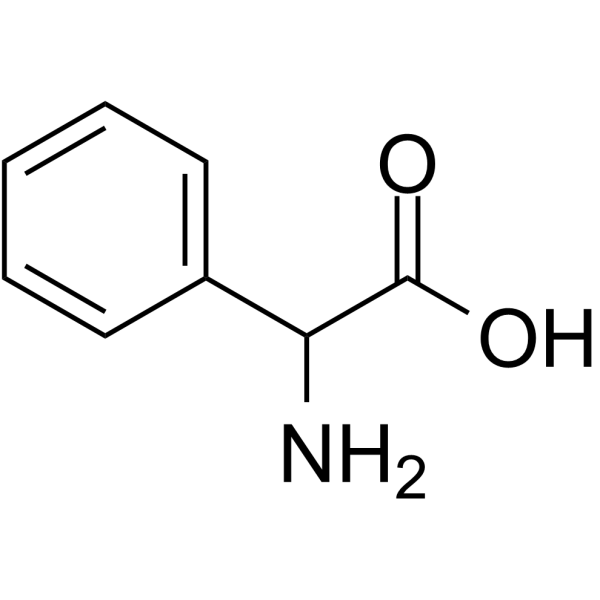
2-Phenylglycine
CAS No. 2835-06-5
2-Phenylglycine( DL-α-Phenylglycine )
Catalog No. M26535 CAS No. 2835-06-5
2-Phenylglycine is a metabolite in breast milk during the lactation period.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 100MG | Get Quote | In Stock |


|
| 200MG | Get Quote | In Stock |


|
| 500MG | 37 | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product Name2-Phenylglycine
-
NoteResearch use only, not for human use.
-
Brief Description2-Phenylglycine is a metabolite in breast milk during the lactation period.
-
Description2-Phenylglycine is a metabolite in breast milk during the lactation period.
-
In Vitro——
-
In Vivo——
-
SynonymsDL-α-Phenylglycine
-
PathwayProteasome/Ubiquitin
-
TargetEndogenous Metabolite
-
RecptorIL-6| p38α
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number2835-06-5
-
Formula Weight151.165
-
Molecular FormulaC8H9NO2
-
Purity>98% (HPLC)
-
SolubilityIn Vitro:?H2O : 3.33 mg/mL (22.03 mM)
-
SMILESNC(C(O)=O)c1ccccc1
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Muchir A, et al. Abnormal p38α mitogen-activated protein kinase signaling in dilated cardiomyopathy caused by lamin A/C gene mutation. Hum Mol Genet. 2012 Oct 1;21(19):4325-33.
molnova catalog



related products
-
D-Ribonolactone
D-Ribonolactone is sugar lactone and an inhibitor of β-galactosidase of Escherichia coli (Ki : 26 Mm).
-
Sphingosine-1-phosph...
Sphingosine-1-phosphate (S1P) is an agonist for S1P1-5 receptors and a ligand for GPR3, GPR6, and GPR12. As an intracellular second messenger, it mobilizes Ca2+ and acts as an extracellular ligand for G-protein coupled receptors.
-
Vasopressin
Vasopressin (argipressin), a cyclic nine-peptide synthesized by hypothalamic neurons, is one of the hormones involved in peptide prohormone synthesis.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


