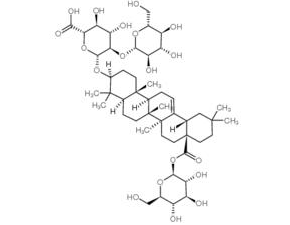
Ginsenoside Ro
CAS No. 34367-04-9
Ginsenoside Ro( Ginsenoside Ro | Chikusetsusaponin 5 )
Catalog No. M18451 CAS No. 34367-04-9
GinsenosideRo exhibits a Ca2+-antagonistic antiplatelet effect.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 1 mL x 10 mM in DMSO | 73 | In Stock |


|
| 5MG | 38 | In Stock |


|
| 10MG | 69 | In Stock |


|
| 25MG | 155 | In Stock |


|
| 50MG | 259 | In Stock |


|
| 100MG | 365 | In Stock |


|
| 200MG | 501 | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameGinsenoside Ro
-
NoteResearch use only, not for human use.
-
Brief DescriptionGinsenosideRo exhibits a Ca2+-antagonistic antiplatelet effect.
-
DescriptionGinsenoside Ro is an inhibitor of autophagosome-lysosome fusion via the ESR2-NCF1-ROS pathway. Ginsenoside Ro also exerted anti-apoptosis and anti-inflammation in IL-1β-induced rat chondrocytes.(In Vitro):Ginsenoside Ro in Panax ginseng is a beneficial novel Ca2+-antagonistic compound and may prevent platelet aggregation-mediated thrombotic disease. Ginsenoside Ro dose-dependently reduces thrombin-stimulated platelet aggregation, and IC50 is approximately 155 μM. Ginsenoside Ro inhibits TXA2 production to abolish thrombin-induced platelet aggregation. Thromboxane A2 (TXA2) induces platelet aggregation and promotes thrombus formation. Ginsenoside Ro dose-dependently (50-300 μM) reduces the TXB2 level that is induced by thrombin; Ginsenoside Ro (300 μM) inhibits the thrombin-mediated elevation in TXB2 level by 94.9%. COX-1 activity in the absence of Ginsenoside Ro (negative control) is 2.3±0.1 nmol/mg protein. However, Ginsenoside Ro dose-dependently (50-300 μM) reduces its activity; at 300 μM, COX-1 activity is reduced by 26.4% of that of the negative control. TXA2 synthase (TXAS) activity in the absence of Ginsenoside Ro (negative control) is 220.8±1.8 ng/mg protein/min. However, Ginsenoside Ro dose-dependently (50-300 μM) reduces its activity; at 300 μM, TXAS activity is reduced by 22.9% of that of the negative control. The inhibitory effect of Ginsenoside Ro (300 μM) on TXB2 production (94.9%) is significantly higher than those on COX-1 (26.4%) and TXAS (22.9%) activities. To assess the toxicity of Ginsenoside Ro in Raw 264.7 cells, they are first treated with various concentrations (10 μM, 50 μM, 100 μM, and 200 μM) of Ginsenoside Ro for 24 h. Ginsenoside Ro exhibits no significant dose dependent toxicity. The effect of Ginsenoside Ro is next determined on cell viability and ROS levels, a marker of oxidative stress, following treatment with 1 μg/mL LPS. LPS reduces cell viability by ~70% compared with nontreated controls. Pretreatment with 100 μM and 200 μM Ginsenoside Ro for 1 h prior to 1 μg/mL LPS incubation for 24 h leads to a significant increase in cell viability. The changes in ROS levels and NO production are consistent with the effects of Ginsenoside Ro on viability. (In Vivo):Ginsenoside Ro dissolved in water is administrated by gavage to mice at doses of 25 and 250 mg/kg/day for 4 days before i.v. injection of HT29 in order to keep blood concentrations of Ginsenoside Ro above a certain level before HT29 i.v. injection followed by 40 days of oral administration of Ginsenoside Ro to the mice. After 38 days of treatment, the animals are euthanized, and the number of pulmonary metastatic nodules is counted in addition to evaluation of toxicity of Ginsenoside Ro and mouse pathology by HT29. Ginsenoside Ro (250 mg/kg/day) produces a significant decrease in the number of tumor nodules on the lung surface, yielding inhibition rates of 88% (P < 0.01).
-
In VitroGinsenoside Ro in Panax ginseng is a beneficial novel Ca2+-antagonistic compound and may prevent platelet aggregation-mediated thrombotic disease. Ginsenoside Ro dose-dependently reduces thrombin-stimulated platelet aggregation, and IC50 is approximately 155?μM. Ginsenoside Ro inhibits TXA2 production to abolish thrombin-induced platelet aggregation. Thromboxane A2 (TXA2) induces platelet aggregation and promotes thrombus formation. Ginsenoside Ro dose-dependently (50-300 μM) reduces the TXB2 level that is induced by thrombin; Ginsenoside Ro (300 μM) inhibits the thrombin-mediated elevation in TXB2 level by 94.9%. COX-1 activity in the absence of Ginsenoside Ro (negative control) is 2.3±0.1 nmol/mg protein. However, Ginsenoside Ro dose-dependently (50-300 μM) reduces its activity; at 300 μM, COX-1 activity is reduced by 26.4% of that of the negative control. TXA2 synthase (TXAS) activity in the absence of Ginsenoside Ro (negative control) is 220.8±1.8 ng/mg protein/min. However, Ginsenoside Ro dose-dependently (50-300 μM) reduces its activity; at 300 μM, TXAS activity is reduced by 22.9% of that of the negative control. The inhibitory effect of Ginsenoside Ro (300 μM) on TXB2 production (94.9%) is significantly higher than those on COX-1 (26.4%) and TXAS (22.9%) activities. To assess the toxicity of Ginsenoside Ro in Raw 264.7 cells, they are first treated with various concentrations (10 μM, 50 μM, 100 μM, and 200 μM) of Ginsenoside Ro for 24 h. Ginsenoside Ro exhibits no significant dose dependent toxicity. The effect of Ginsenoside Ro is next determined on cell viability and ROS levels, a marker of oxidative stress, following treatment with 1 μg/mL LPS. LPS reduces cell viability by ~70% compared with nontreated controls. Pretreatment with 100 μM and 200 μM Ginsenoside Ro for 1 h prior to 1 μg/mL LPS incubation for 24 h leads to a significant increase in cell viability. The changes in ROS levels and NO production are consistent with the effects of Ginsenoside Ro on viability.
-
In VivoGinsenoside Ro dissolved in water is administrated by gavage to mice at doses of 25 and 250?mg/kg/day for 4 days before i.v. injection of HT29 in order to keep blood concentrations of Ginsenoside Ro above a certain level before HT29 i.v. injection followed by 40 days of oral administration of Ginsenoside Ro to the mice. After 38 days of treatment, the animals are euthanized, and the number of pulmonary metastatic nodules is counted in addition to evaluation of toxicity of Ginsenoside Ro and mouse pathology by HT29. Ginsenoside Ro (250?mg/kg/day) produces a significant decrease in the number of tumor nodules on the lung surface, yielding inhibition rates of 88% (P?SynonymsGinsenoside Ro | Chikusetsusaponin 5PathwayNeuroscienceTargetGluRRecptor5-α reductaseResearch AreaOthers-FieldIndication——
Chemical Information
-
CAS Number34367-04-9
-
Formula Weight957.11
-
Molecular FormulaC48H76O19
-
Purity>98% (HPLC)
-
SolubilityDMSO : 100 mg/mL 104.48 mM
-
SMILESO1[C@@H]([C@H]([C@@H]([C@H]([C@@H]1O[C@H]1CC[C@]2([C@H](C1(C)C)CC[C@]1([C@@]3(CC[C@]4(CCC(C[C@H]4C3=CC[C@H]21)(C)C)C(=O)O[C@@H]1O[C@@H]([C@H]([C@@H]([C@H]1O)O)O)CO)C)C)C)O[C@@H]1O[C@@H]([C@H]([C@@H]([C@H]1O)O)O)CO)O)O)C(=O)O
-
Chemical Name(2S,3S,4S,5R,6R)-6-[[(3S,4aR,6aR,6bS,8aS,12aS,14aR,14bR)-4,4,6a,6b,11,11,14b-Heptamethyl-8a-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxycarbonyl-1,2,3,4a,5,6,7,8,9,10,12,12a,14,14a-tetradecahydropicen-3-yl]oxy]-3,4-dihydroxy-5-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyoxane-2-carboxylic acid
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1. Kang HJ, et al. Antioxidative properties of ginsenoside Ro against UV-B-induced oxidative stress in human dermal fibroblasts. Biosci Biotechnol Biochem. 2015 Jul 27:1-4.
molnova catalog



related products
-
Tebanicline hydrochl...
Tebanicline (ABT-594, Ebanicline), is an effective synthetic nicotinic (non-opioid) analgesic drug.
-
Motolimod
Motolimod (VTX-2337) is an effective and specific Toll-like receptor (TLR) 8 agonist (EC50: 100 nM), > 50-fold selectivity over TLR7.
-
CPPHA
CPPHA, a positive allosteric modulator of glutamate receptors mGluR5 and mGluR1, is commonly used in development for central nervous system diseases.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


