CCT-367766
CAS No. 2229856-58-8
CCT-367766( CCT367766 | CCT 367766 )
Catalog No. M13570 CAS No. 2229856-58-8
CCT-367766 (CCT367766) is a novel heterobifunctional PROTAC that binds and degrades the putative transcription factor regulator Pirin in cells.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 5MG | 686 | In Stock |


|
| 10MG | 938 | In Stock |


|
| 25MG | 1444 | In Stock |


|
| 50MG | 1841 | In Stock |


|
| 100MG | Get Quote | In Stock |


|
| 200MG | Get Quote | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameCCT-367766
-
NoteResearch use only, not for human use.
-
Brief DescriptionCCT-367766 (CCT367766) is a novel heterobifunctional PROTAC that binds and degrades the putative transcription factor regulator Pirin in cells.
-
DescriptionCCT-367766 (CCT367766) is a novel heterobifunctional PROTAC that binds and degrades the putative transcription factor regulator Pirin in cells.
-
In VitroCCT367766 (50-1500 nM; 24 hours) demonstates the depletion of pirin protein as a?the time-dependent hook-effect in SK-OV-3 human ovarian cancer cells.CCT367766 (0.5-50 nM; 2 hours) demonstrates the concentration-dependent depletion of pirin protein after 2 h exposure in SK-OV-3 cells.CCT367766 (0.5-50 nM; 2 hours) dose-dependently rescuses pirin expression from pretreatment of chlorobisamide in SK-OV-3 cells.Western Blot AnalysisCell Line:SK-OV-3 human ovarian cancer cells Concentration:50, 150, 250, 500 and 1500 nMIncubation Time:2 hours, 4 hours, 24 hours Result:Decreased pirin protein expression.Western Blot Analysis Cell Line:SK-OV-3 human ovarian cancer cells Concentration:0.5, 1, 2.5, 5, 7.5, 10, 25, and 50 nM Incubation Time:2 hours Result:Completely degraded pirin just at50 nM treatment.
-
In Vivo——
-
SynonymsCCT367766 | CCT 367766
-
PathwayPROTACs
-
TargetPROTAC
-
RecptorPROTAC
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number2229856-58-8
-
Formula Weight946.41
-
Molecular FormulaC49H48ClN7O11
-
Purity>98% (HPLC)
-
Solubility——
-
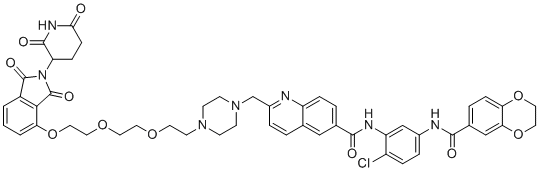
SMILESO=C(C1=CC=C2N=C(CN3CCN(CCOCCOCCOC4=CC=CC(C(N5C(CC6)C(NC6=O)=O)=O)=C4C5=O)CC3)C=CC2=C1)NC7=CC(NC(C8=CC=C9OCCOC9=C8)=O)=CC=C7Cl
-
Chemical NameN-(2-Chloro-5-(2,3-dihydrobenzo[b][1,4]dioxine-6-carboxamido)phenyl)-2-((4-(2-(2-(2-((2-(2,6-dioxopiperidin-3-yl)-1,3-dioxoisoindolin-4-yl)oxy)ethoxy)ethoxy)ethyl)piperazin-1-yl)methyl)quinoline-6-carboxamide
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1. Chessum NEA, et al. J Med Chem. 2017 Dec 14. doi: 10.1021/acs.jmedchem.7b01406.
molnova catalog



related products
-
BRD4 degrader AT1
BRD4 degrader AT1 is a highly selective BED4 degrader (PROTAC), exhibits highly selective depletion of BRD4 in cells with negligible activity against BRD2 and BRD3.
-
Hydroxy-PEG6-Boc
Hydroxy-PEG7-Boc is a PEG-based PROTAC linker that can be used in the synthesis of PROTACs.
-
ZXH-3-26
ZXH-3-26 is a novel BRD4 heterobifunctional small-molecule ligand (PROTAC), shows activity exclusively on the BRD4 BD1 (DC50/5h=5 nM) and spares degradation of BRD2 or BRD3 in cellular degradation assays.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


