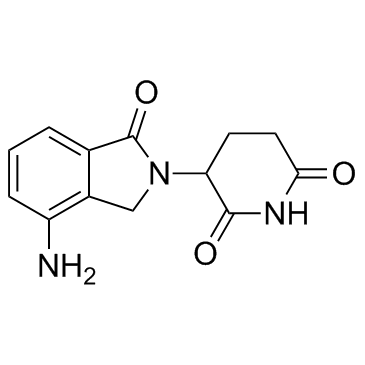
Lenalidomide
CAS No. 191732-72-6
Lenalidomide ( CC-5013 )
Catalog No. M13005 CAS No. 191732-72-6
Lenalidomide (initially known as CC-5013 and marketed as Revlimid by Celgene) is a derivative of thalidomide introduced in 2004.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 1 mL x 10 mM in DMSO | 48 | In Stock |


|
| 50MG | 33 | In Stock |


|
| 100MG | 45 | In Stock |


|
| 200MG | 54 | In Stock |


|
| 500MG | 67 | In Stock |


|
| 1G | 91 | In Stock |


|
Biological Information
-
Product NameLenalidomide
-
NoteResearch use only, not for human use.
-
Brief DescriptionLenalidomide (initially known as CC-5013 and marketed as Revlimid by Celgene) is a derivative of thalidomide introduced in 2004.
-
DescriptionLenalidomide (initially known as CC-5013 and marketed as Revlimid by Celgene) is a derivative of thalidomide introduced in 2004. It was initially intended as a treatment for multiple myeloma, for which thalidomide is an accepted therapeutic modality, but has also shown efficacy in the hematological disorders known as the myelodysplastic syndromes. FDA approved on December 27, 2005.(In Vitro):Lenalidomide is potent in stimulating T cell proliferation and IFN-γ and IL-2 production. Lenalidomide has been shown to inhibit production of pro inflammatory cytokines TNF-α, IL-1, IL-6, IL-12 and elevate the production of anti-inflammatory cytokine IL-10 from human PBMCs. Lenalidomide downregulates the production of IL-6 directly and also by inhibiting multiple myeloma (MM) cells and bone marrow stromal cells (BMSC) interaction, which augments the apoptosis of myeloma cells. Dose-dependent interaction with the CRBN-DDB1 complex is observed with Thalidomide, Lenalidomide and Pomalidomide, with IC50 values of ~30 μM, ~3 μM and ~3 μM, respectively, These reduced CRBN expression cells (U266-CRBN60 and U266-CRBN75) are less responsive than the parental cells to antiproliferative effects Lenalidomide across a dose-response range of 0.01 to 10 μM. Lenalidomide, a thalidomide analog, functions as a molecular glue between the human E3 ubiquitin ligase cereblon and CKIα is shown to induce the ubiquitination and degradation of this kinase, thus presumably killing leukemic cells by p53 activation.(In Vivo):The toxicity of Lenalidomide doses up to 15, 22.5, and 45 mg/kg via IV, IP, and PO routes of administration. Limited by solubility in our PBS dosing vehicle, these maximum achievable Lenalidomide doses are well tolerated with the exception of one mouse death (of four total dosed) at the 15 mg/kg IV dose. Notably, no other toxicities are observed in the study at IV doses of 15 mg/kg (n=3) or 10 mg/kg (n=45) or at any other dose level through IV, IP, and PO routes.
-
In VitroLenalidomide is potent in stimulating T cell proliferation and IFN-γ and IL-2 production. Lenalidomide has been shown to inhibit production of pro inflammatory cytokines TNF-α, IL-1, IL-6, IL-12 and elevate the production of anti-inflammatory cytokine IL-10 from human PBMCs. Lenalidomide downregulates the production of IL-6 directly and also by inhibiting multiple myeloma (MM) cells and bone marrow stromal cells (BMSC) interaction, which augments the apoptosis of myeloma cells. Dose-dependent interaction with the CRBN-DDB1 complex is observed with Thalidomide, Lenalidomide and Pomalidomide, with IC50 values of ~30 μM, ~3 μM and ~3 μM, respectively, These reduced CRBN expression cells (U266-CRBN60 and U266-CRBN75) are less responsive than the parental cells to antiproliferative effects Lenalidomide across a dose-response range of 0.01 to 10 μM. Lenalidomide, a thalidomide analog, functions as a molecular glue between the human E3 ubiquitin ligase cereblon and CKIα is shown to induce the ubiquitination and degradation of this kinase, thus presumably killing leukemic cells by p53 activation.
-
In VivoThe toxicity of Lenalidomide doses up to 15, 22.5, and 45 mg/kg via IV, IP, and PO routes of administration. Limited by solubility in our PBS dosing vehicle, these maximum achievable Lenalidomide doses are well tolerated with the exception of one mouse death (of four total dosed) at the 15 mg/kg IV dose. Notably, no other toxicities are observed in the study at IV doses of 15 mg/kg (n=3) or 10 mg/kg (n=45) or at any other dose level through IV, IP, and PO routes.
-
SynonymsCC-5013
-
PathwayApoptosis
-
TargetTNF
-
RecptorTNF-α
-
Research AreaCancer
-
Indication——
Chemical Information
-
CAS Number191732-72-6
-
Formula Weight259.26
-
Molecular FormulaC13H13N3O3
-
Purity>98% (HPLC)
-
SolubilityDMSO: ≥ 35 mg/mL
-
SMILESO=C(C(N(CC1=C2C=CC=C1N)C2=O)CC3)NC3=O
-
Chemical Name3-(4-amino-1-oxoisoindolin-2-yl)piperidine-2,6-dione
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Muller GW, et al. Bioorg Med Chem Lett, 1999, 9(11), 1625-1630.
molnova catalog



related products
-
INH1
INH1 is a cell-permeable Hec1 inhibitor that specifically disrupts the Hec1/Nek2 interaction.
-
Aflatoxin G1
Aflatoxin G1 is one type of aflatoxins occuring in nature. It is produced by molds, such as Aspergillus flavus and Aspergillus parasiticus. Aflatoxins are hepatogenic, teratogenic, imunosuppressive, and carcinogenic fungal metabolites found in feeds, nuts, wine-grapes, spices, and other grain crops.
-
Brentuximab
Brentuximab, a chimeric antibody targeting CD30, is a naked antibody to Brentuximab vedotin. It possesses antitumor activity and may be used to study relapsed or refractory Hodgkin's lymphoma.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


