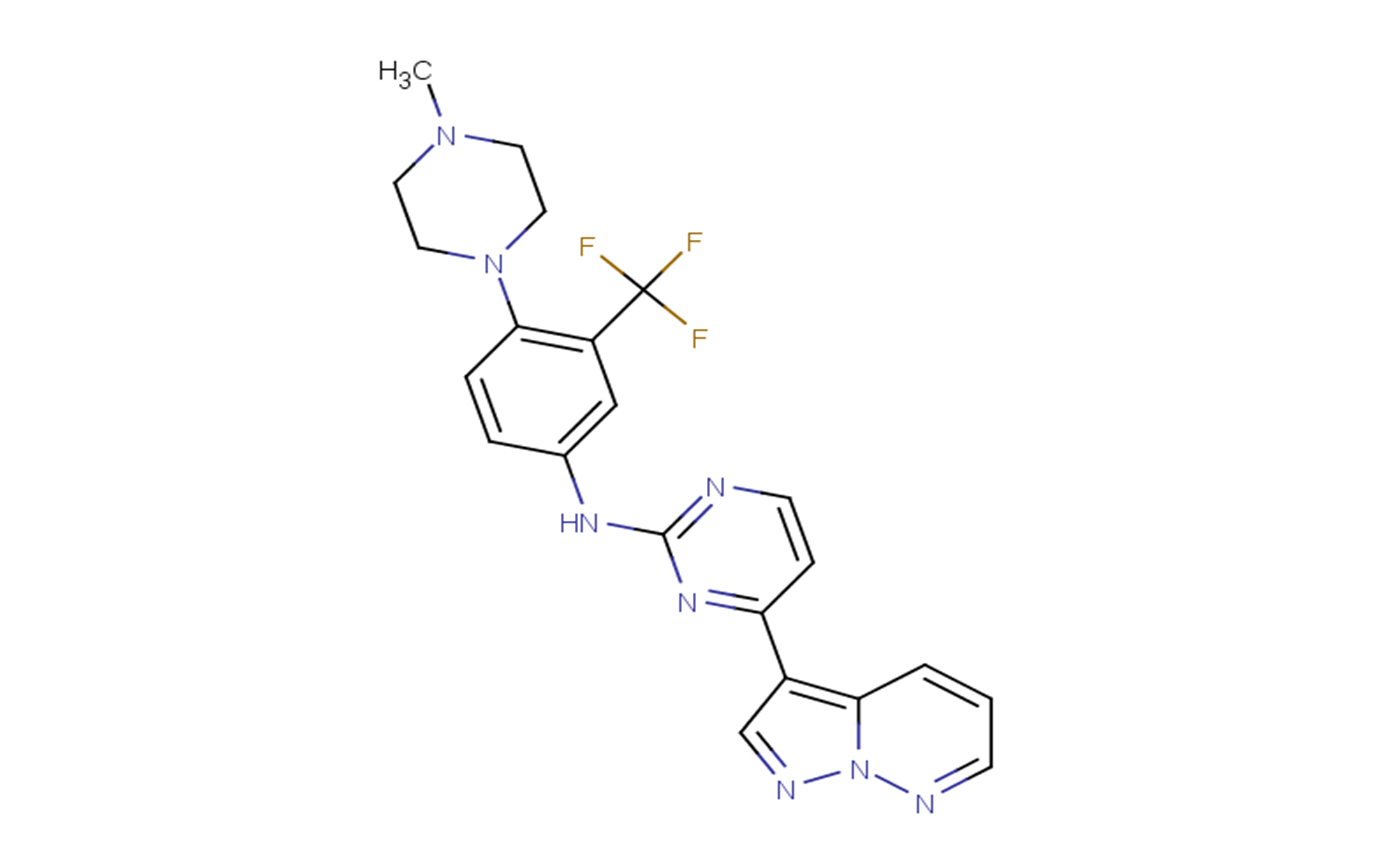
GW779439X
CAS No. 551919-98-3
GW779439X( —— )
Catalog No. M24539 CAS No. 551919-98-3
GW779439X is an inhibitor of CDK.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 1 mL x 10 mM in DMSO | 126 | In Stock |


|
| 2MG | 75 | In Stock |


|
| 5MG | 116 | In Stock |


|
| 10MG | 188 | In Stock |


|
| 25MG | 426 | In Stock |


|
| 50MG | 612 | In Stock |


|
| 100MG | 872 | In Stock |


|
| 200MG | Get Quote | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameGW779439X
-
NoteResearch use only, not for human use.
-
Brief DescriptionGW779439X is an inhibitor of CDK.
-
DescriptionGW779439X is an inhibitor of CDK.
-
In Vitro——
-
In Vivo——
-
Synonyms——
-
PathwayAngiogenesis
-
TargetCDK
-
RecptorCDK
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number551919-98-3
-
Formula Weight454.45
-
Molecular FormulaC22H21F3N8
-
Purity>98% (HPLC)
-
SolubilityDMSO:10 mM
-
SMILESCN1CCN(CC1)C2=C(C=C(C=C2)NC3=NC=CC(=N3)C4=C5C=CC=NN5N=C4)C(F)(F)F
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Schaenzer A J , Wlodarchak N , Drewry D H , et al. GW779439X and Its Pyrazolopyridazine Derivatives Inhibit the Serine/Threonine Kinase Stk1 and Act As Antibiotic Adjuvants against β-Lactam-Resistant Staphylococcus aureus[J]. other, 2018, 4(10).
molnova catalog



related products
-
TBB
TBB(NSC 231634) is a highly selective, ATP/GTP-competitive inhibitor of casein kinase-2 (CK2)with IC50s of 0.9 and 1.6 μM for rat liver and human recombinant CK2 respectively).
-
CDK7 and 9 inhibitor...
A potent, selective transcriptional CDK inhibitor with Ki of 2.3 and 0.38 nM for CDK7 and CDK9, respectively; displays >30-fold selectivity over CDK1/2/4, inhibits VEGFR2 with IC50 of 180 nM in a panel of non-CDK kinases.
-
SEL120-34A
SEL120-34A is an ATP-competitive, selective, orally active CDK8 inhibitor that inhibits kinase activities of CDK8/CycC and CDK19/CycC complexes with IC50 of 4.4 nM and 10.4 nM, respectively.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


