PKR-IN-2
CAS No. 1628428-01-2
PKR-IN-2( —— )
Catalog No. M23724 CAS No. 1628428-01-2
PKR-IN-2 is a activator of pyruvate kinase (PKR).
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 1 mL x 10 mM in DMSO | 144 | In Stock |


|
| 5MG | 141 | In Stock |


|
| 10MG | 207 | In Stock |


|
| 25MG | 334 | In Stock |


|
| 50MG | 457 | In Stock |


|
| 100MG | 642 | In Stock |


|
| 200MG | 864 | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NamePKR-IN-2
-
NoteResearch use only, not for human use.
-
Brief DescriptionPKR-IN-2 is a activator of pyruvate kinase (PKR).
-
DescriptionPKR-IN-2 is a activator of pyruvate kinase (PKR).
-
In Vitro——
-
In Vivo——
-
Synonyms——
-
PathwayOthers
-
TargetOther Targets
-
Recptorpyruvate kinase
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number1628428-01-2
-
Formula Weight468.57
-
Molecular FormulaC24H28N4O4S
-
Purity>98% (HPLC)
-
SolubilityDMSO:45mg/mL (96.04 mM; Need ultrasonic)
-
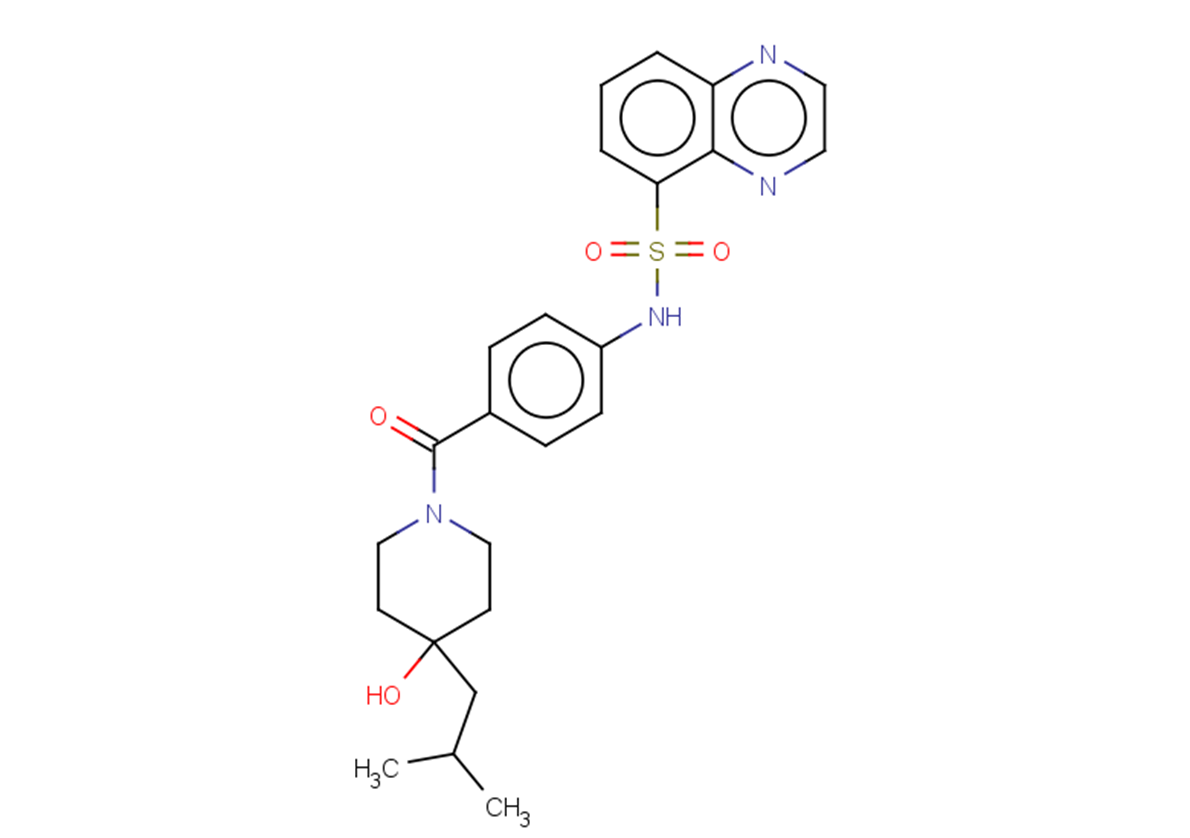
SMILESCC(C)CC(CC1)(CCN1C(c(cc1)ccc1NS(c1cccc2nccnc12)(=O)=O)=O)O
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Popovici-Muller, et al. Preparation of N-(piperidinecarbonylphenyl) sulfonamide compounds as pyruvate kinase modulators. From PCT Int. Appl. (2014), WO2014139144A1 20140918.
molnova catalog



related products
-
Beryllon II
Beryllon II is a widely used chromogenic reagent that is used to determine proteins and many elements, such as Mg, Mo, and Co.
-
Erythrosine
A tetraiodofluorescein used as a red coloring in some foods (cherries, fish), as a disclosure of DENTAL PLAQUE, and as a stain of some cell types. It has structural similarity to THYROXINE.
-
Ziconotide TFA (1074...
Ziconotide TEA acts by binding to N-type calcium channels situated on the terminal part of primary afferent neurons of the nociceptive pathway therefore reducing synaptic transmission with potent antinociceptive effects.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


