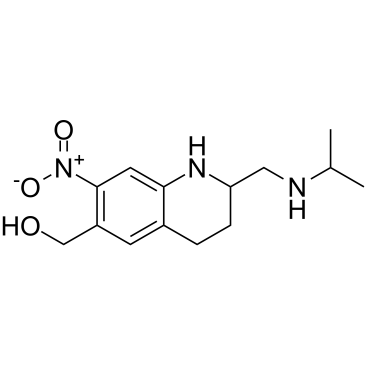
Oxamniquine
CAS No. 21738-42-1
Oxamniquine ( —— )
Catalog No. M21872 CAS No. 21738-42-1
Oxamniquine is a potent agent for the treatment of schistosomiasis.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 5MG | 1143 | Get Quote |


|
| 50MG | 2322 | Get Quote |


|
| 100MG | 3060 | Get Quote |


|
| 200MG | Get Quote | Get Quote |


|
| 500MG | Get Quote | Get Quote |


|
| 1G | Get Quote | Get Quote |


|
Biological Information
-
Product NameOxamniquine
-
NoteResearch use only, not for human use.
-
Brief DescriptionOxamniquine is a potent agent for the treatment of schistosomiasis.
-
DescriptionOxamniquine is a potent agent for the treatment of schistosomiasis.
-
In VitroOxamniquine is a potent agent for the treatment of schistosomiasis.
-
In VivoOxamniquine (500 mg/kg) alone or Oxamniquine (250 mg/kg) in combination with praziquantel reduces the mortality of infected snails compared to the control group.
-
Synonyms——
-
PathwayMicrobiology/Virology
-
TargetParasite
-
RecptorParasite
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number21738-42-1
-
Formula Weight279.33
-
Molecular FormulaC??H??N?O?
-
Purity>98% (HPLC)
-
Solubility——
-
SMILESOCC1=C([N+]([O-])=O)C=C2C(CCC(CNC(C)C)N2)=C1
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1. Mattos AC, et al. Evaluation of the effect of oxamniquine, praziquantel and a combination of both drugs on the intramolluscan phase of Schistosoma mansoni. Acta Trop. 2007 May;102(2):84-91.
molnova catalog



related products
-
N-(2-Hydroxypropyl)m...
N-(2-Hydroxypropyl)methacrylamide is used in the synthesis of copolymers for the targeted delivery of antileishmanial agents in Visceral leishmaniasis.
-
YCN47284
YCN47284 is an antimalarial compound with an IC50 of 1 μM for P. falciparum.
-
Naphthoquine phospha...
An antimalarial drug.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


