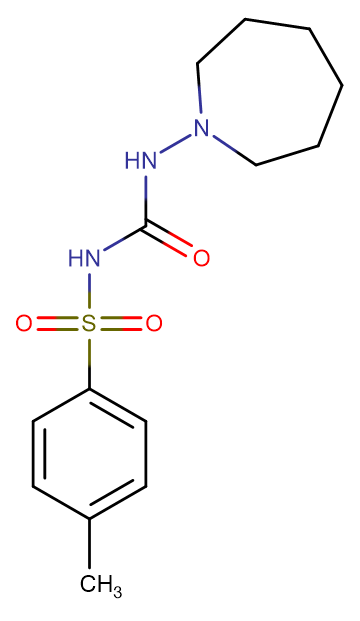
Tolazamide
CAS No. 1156-19-0
Tolazamide( NSC 70762 )
Catalog No. M10547 CAS No. 1156-19-0
Tolazamide is an oral blood glucose lowering drug used for people with Type 2 diabetes.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 1 mL x 10 mM in DMSO | 53 | In Stock |


|
| 2MG | 29 | In Stock |


|
| 5MG | 46 | In Stock |


|
| 10MG | 61 | In Stock |


|
| 25MG | 93 | In Stock |


|
| 50MG | 118 | In Stock |


|
| 100MG | 152 | In Stock |


|
| 200MG | 226 | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameTolazamide
-
NoteResearch use only, not for human use.
-
Brief DescriptionTolazamide is an oral blood glucose lowering drug used for people with Type 2 diabetes.
-
DescriptionTolazamide is an oral blood glucose lowering drug used for people with Type 2 diabetes.
-
In Vitro——
-
In Vivo——
-
SynonymsNSC 70762
-
PathwayCell Cycle/DNA Damage
-
TargetPotassium Channel
-
RecptorPotassium Channel
-
Research AreaEndocrinology
-
Indication——
Chemical Information
-
CAS Number1156-19-0
-
Formula Weight311.4
-
Molecular FormulaC14H21N3O3S
-
Purity>98% (HPLC)
-
SolubilityWater: 65.4 mg/L (at 30 °C)
-
SMILESO=C(NS(=O)(C1=CC=C(C)C=C1)=O)NN2CCCCCC2
-
Chemical Name1-(azepan-1-yl)-3-(4-methylphenyl)sulfonylurea
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Szabo C, Salzman AL. Shock. 1996 Jun;5(6):391-4.
molnova catalog



related products
-
Pinacidil
Pinardil (P-1134, S-1230) is a guanidine that opens potassium channels and directly dilates peripheral blood vessels in small arteries, lowering blood pressure and peripheral resistance and producing fluid retention.
-
MK-0448
MK-0448 is a potent, specific I(Kur) current/Kv1.5 channel inhibitor with IC50 of 8.6 nM, potently inhibits IKur in human atrial myocytes with IC50 of 10.8 nM.
-
TRAM 39
TRAM 39 is a selective blocker of intermediate-conductance Ca2+-activated K+channels.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


