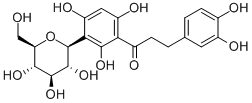
Aspalathin
CAS No. 6027-43-6
Aspalathin( —— )
Catalog No. M29643 CAS No. 6027-43-6
Aspalathin is a C-linked dihydrochalcone glucoside found in rooibos tea, a herbal tea prepared from the South African rooibos plant, Aspalathus linearis (Fabaceae).
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 100MG | Get Quote | In Stock |


|
| 200MG | Get Quote | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameAspalathin
-
NoteResearch use only, not for human use.
-
Brief DescriptionAspalathin is a C-linked dihydrochalcone glucoside found in rooibos tea, a herbal tea prepared from the South African rooibos plant, Aspalathus linearis (Fabaceae).
-
DescriptionAspalathin is a C-linked dihydrochalcone glucoside found in rooibos tea, a herbal tea prepared from the South African rooibos plant, Aspalathus linearis (Fabaceae).
-
In Vitro——
-
In Vivo——
-
Synonyms——
-
PathwayOthers
-
TargetOther Targets
-
Recptor——
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number6027-43-6
-
Formula Weight452.4
-
Molecular FormulaC21H24O11
-
Purity>98% (HPLC)
-
SolubilitySoluble in methanol and water
-
SMILESC1=CC(=C(C=C1CCC(=O)C2=C(C(=C(C=C2O)O)C3C(C(C(C(O3)CO)O)O)O)O)O)O
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
molnova catalog



related products
-
Chlorophyllin
Chlorophyllin sodium copper salt is a semi-synthetic mixture of water-soluble sodium copper salts derived from chlorophyll.
-
2-Aminopurine-9-beta...
2-Aminopurine-9-beta-D-(2’-deoxy)riboside, a derivative of 2-Aminopurine, is a modified nucleoside composed of a purine base linked to a (2’-deoxy)ribose sugar moiety.
-
Ecliptasaponin A
Ecliptasaponin A has protective effects against the pulmonary fibrosis induced by bleomycin via reducing the oxidative stress, lung tissue inflammation, and the subsequent epithelial-mesenchymal transition.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


